REGULATORY FOCUS: FEED CLASSIFICATION
🔍 Sarasvet Regulatory Focus : February 2026 | Part 3
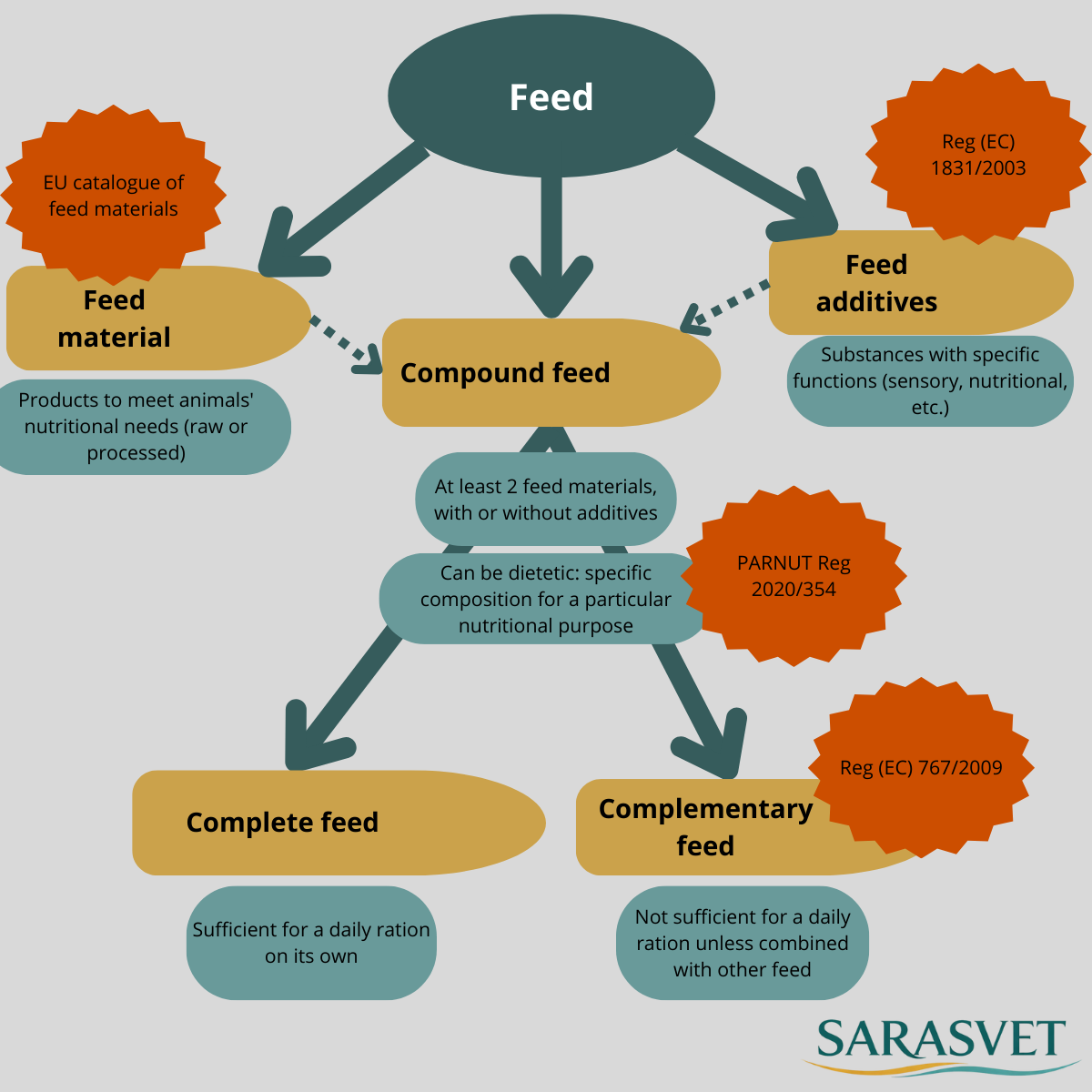
Complete feed, complementary feed, dietetic feed… do we all speak the same regulatory language? 🤔
In animal nutrition, categories are strictly defined in EU law. Confusing them can quickly lead to non-compliant claims or product reclassification. Here’s a clear and practical overview (EU context) 👇
🍽️ Complete feed (animals)
- Covers all daily nutritional needs
- Can be fed as the sole ration
➕ Complementary feed (animals)
- Must be used in combination with other feed to ensure the daily ration
- Defined under Regulation (EC) No 767/2009
🩺 Dietetic feed / PARNUT (Feed intended for PARticular NUTritional purposes)
- Can be complete or complementary
- Targets a specific nutritional objective (e.g. renal support, digestion)
- Strictly governed by Regulation (EU) 2020/354
🧪 Feed additives (animals)
- Substances (vitamins, enzymes, etc.) added for a specific function
- Authorised under Regulation (EC) No 1831/2003
🌾 Feed materials (animals)
- Single ingredients used to formulate feeds
- Referenced in the EU Catalogue of feed materials
⚠️ Feed vs. Medicine: the grey zone
- A feed (even a dietetic one) is never medicinal. If a product claims to prevent, treat, or cure a disease, it falls under Veterinary Medicine (Regulation (EU) 2019/6). This is the biggest risk for companies
💊 Medicated Feed
- Mixture of veterinary medicine and feed, subject to veterinary prescription
- Medicated Premix: an authorised veterinary medicine designed to be incorporated into feed
- Regulation (EU) 2019/4
🚫 Food supplements (humans)
- A human-only regulatory category
- ❌ Does not exist in animal feed legislation. In the animal world, we speak of additives or complementary feeds.
🎯 Take-home message
In animal health, correct classification drives your claims, evidence requirements and market access.